MultNAT® STI Panel (14-plex)

Product Overview
MultNAT STI Panel is an automated, in vitro nucleic acid amplification test designed for the qualitative detection and differentiation of 14 sexually transmitted infection (STI) pathogens in urine and female genital swab specimens. It provides quantitative information for Ureaplasma urealyticum (UU), Mycoplasma hominis (MH), and Gardnerella vaginalis (GV) to support diagnosis and treatment. The assay aids in the diagnosis of STIs including Chlamydia, Gonorrhea, Syphilis, Trichomoniasis, Genital Herpes, Chancroid, Non-gonococcal Urethritis, and Vaginitis.
- Fast & Integrated: From sample to result in 66 minutes with fully automated “sample-in, result-out” workflow
- Quantitative Insight:Provides bacterial load for UU, MH, and GV to support treatment decisions
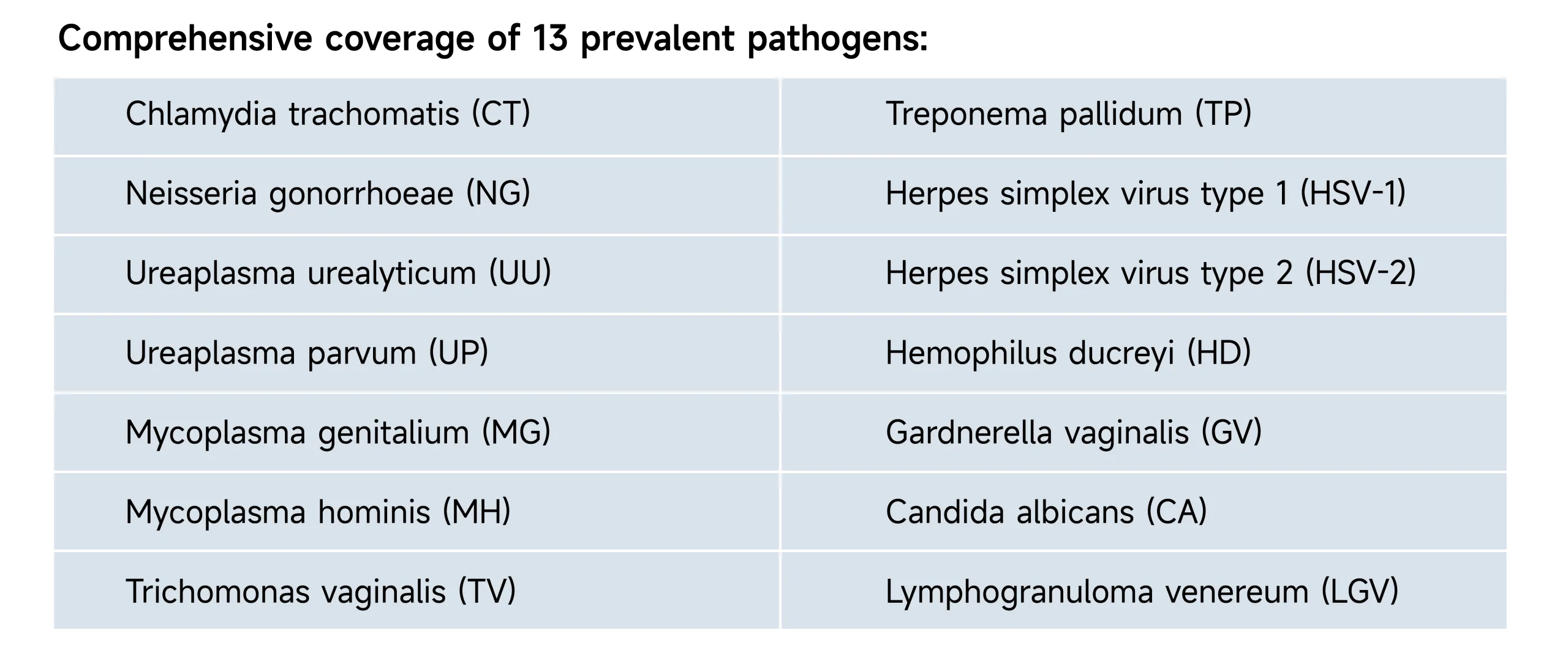
- Comprehensive Coverage: Detects CT, NG, TV, TP, HSV-1/2, UU, UP, MH, MG, GV, HD, CA, and LGV serovars in one run
- Closed & Contamination-Free: All-in-one cartridge design eliminates cross-contamination risk
Ongoing Challanges
Sexually transmitted infections (STIs) present a growing global health burden, with over 374 million new curable cases annually (WHO 2020). Yet traditional detection methods often fall short due to:
Multiple Pathogens, Single Sample: Symptoms overlap, but tests are often pathogen-specific
Asymptomatic Carriers: Up to 70% of infections show no symptoms, yet can lead to serious complications
Complex Workflows: Manual steps increase turnaround time and contamination risk
Quantitative Gaps: Some pathogens require bacterial load data to guide therapy
Clinical Value
- Comprehensive Detection: Covers 14 key STI pathogens in a single test
- Quantitative Capability: Provides bacterial load data for UU, MH, and GV to guide treatment
- High Throughput: 4/8 tests per run and compatible with the MultNAT Molecular Diagnostic Testing System for efficient batch processing
- Robust Performance: Validated for both symptomatic and asymptomatic individuals