Products
Diagnostic Kit for Novel-Coronavirus (2019-nCov) RNA (PCR-Fluorescent Probe Assay)
- Categories:PCR Reagent
- Time of issue:2020-10-12 13:52:23
- Views:0
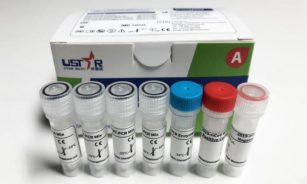
【Introduction】
This reagent uses one-step reverse transcription polymerase chain reaction (one step RT-PCR) combined with Taqman probe technology. Primers and probes are designed based on the WHO’s latest recommended gene fragments. It is suitable for in vitro qualitative detection of novel coronavirus (2019-nCoV) ribonucleic acid (RNA) in human oro[haryngeal swabs, nasopharyngeal swabs, sputum and other samples.
【Advantages】
● Panel testing
● High efficiency
● High sensitivity
● High specificity
● High accuracy
● Wide applicability
【Specification】
200 tests/kit
【Applicable Equipment】
Abbott m2000rt RealTime PCR System
Contact us
Ustar Biotechnologies (Hangzhou) Ltd. (Headquarters)
Add: 6/F, Building 2, 611 Dongguan Road, Binjiang District, Hangzhou, Zhejiang, P.R. China
Zip: 310053
Email: sales@bioustar.com service@bioutsar.com
Tel: +86-571-88939358 (Sales)
+86-400-870-7025 (Service)
Copyright Ustar Biotechnologies 2018. All rights reserved 浙ICP备20029265号




